While we will focus here on MS and discuss some new research that has recently been published, I believe that given the interaction of so many factors in the treatment of any neurological disease, it’s important to look for all the drivers of systemic and neuro-inflammation. In the case of MS, there are numerous mechanisms that essentially opened the door to disease initiation that can direct the course of the disease and impact the success or failure of a particular treatment. So while offering immune-modulating therapies, for example, may be helpful in some patients, there are times these approaches are just not effective. This is not necessarily because a specific drug isn’t working; rather, the progression of MS in each patient is as diverse as that individual. And so, the disease can be fueled by these respective agendas that both the brain and body have manifested that are themselves dictated by such factors as other health issues, environmental influences, exposure to toxins, diet and more.
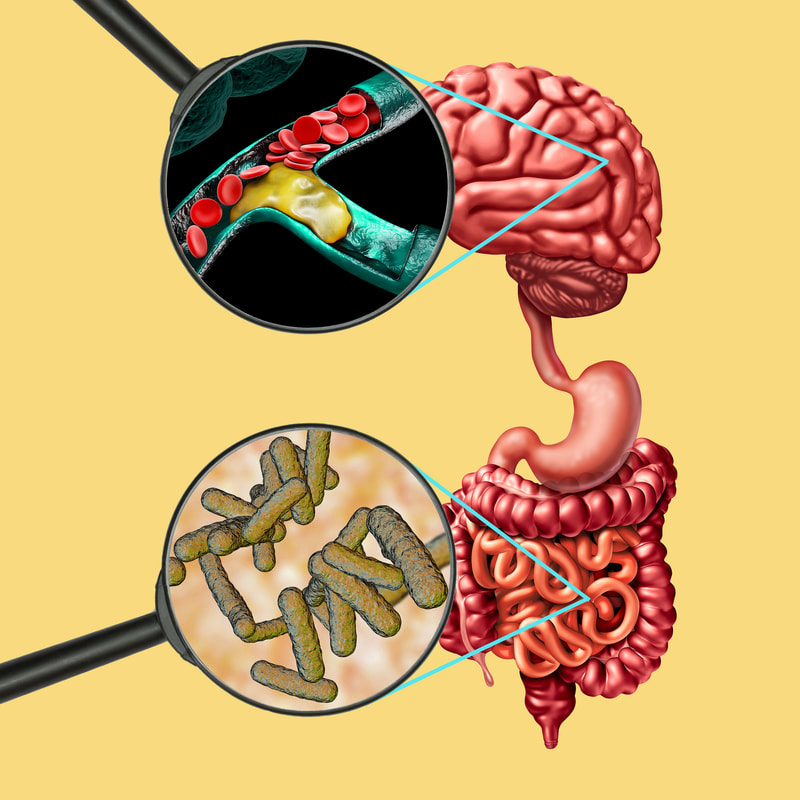
Let’s look at the research. People with MS are at a greater risk of macrovascular events (damage to large blood vessels), all-cause mortality, and mortality related to cardiovascular issues, according to a large population study recently published in JAMA Neurology. Other studies have suggested that vascular abnormalities associated with the disease may actually play a role in its pathogenesis (the manner in which it develops). In this recent study, data was derived from more than 12,000 people with MS, matched by commensurate study controls. According to researchers the findings shed a glaring light on the cardiovascular risks that MS patients face and “point to the need for careful assessment of these risks in order to optimize management of all MS patients.” The findings also underscored the heightened risks of women with MS who fared worse on most of the outcomes including 30% higher vascular risks in female patients with MS compared to those without the illness. Clearly these findings indicate that multiple sclerosis is associated with an increased risk of cardiovascular and cerebrovascular disease for which traditional risk factors cannot completely account. Mechanisms of Vascular Comorbidity in MS. Despite significant advances in treatment, patients with MS die significantly earlier than the general population because of cardiovascular comorbidities, mostly in connection with accelerated atherosclerosis. In integrative medicine, we believe that INFLAMMATION is at the root of every disease. Could factors driving high levels of inflammation not only be the root cause of MS, but also what is driving the increased risk of vascular disease? Together with the loss of myelin and the glial proliferation, the perivascular infiltration and endothelial damage have been described as histological findings in the brain tissue of patients with MS. In other words, the brain and the brain’s vessels are very inflamed. What fuels inflammation? In our unfortunately highly toxic world today, it is rarely one thing that leads to disease of any kind. It’s almost “the perfect storm” that represents a rising tide of contributors, such as: 1. Altered microbiome In 2001, Nobel Prize winner Joshua Lederberg coined the term “microbiome.” The microbiome is the “ecological community of commensal, symbiotic and pathogenic microorganisms” that can be found on mucosal surfaces, including the eye, mouth, lungs, and the gut. There is much research linking MS with intestinal permeability. Additional investigations show a link between the microbiome and pediatric MS, suggesting a connection between myelin production and metabolites produced by gut microbes (particularly p-cresol). Short chain fatty acids from the bowel flora (especially butyrate) affect remyelination, microglia function, and also oligodendrocyte differentiation. In the autoimmune disease, neuromyelitis optica, research also shows a potential connection to bowel ecology. Yet another study identified a connection between central nervous system (CNS) inflammation and the gut microbiome in mice. So, there is no disputing the gut-brain and, conversely, the brain-gut connection. An increase in the permeability of the intestinal mucosa, which could allow bacteria, toxic digestive metabolites, bacterial toxins, and small molecules to leak into the bloodstream is known as leaky gut. And a leaky gut from gut dysbiosis, or microbial imbalance, is associated with leaky brain.1 So, when we consider the cause of neurological disease initiation, we also have to consider this relationship. The microbiota-gut is an integral component of the gut-brain neuroendocrine metabolic axis and any disruption that occurs (e.g. antibiotic use for infections, gluten consumption, unhealthy diets, exposure to environmental chemicals, toxins, pesticides such a glyphosates found prayed on nonorganic food, etc.), subsequently can upset homeostasis as well as share an inflammatory component. The gut can influence the blood-brain barrier (BBB), a single-layered lattice of cells joined by tight junctions that keep out most substances from entering the brain. However, by means of gastrointestinal-derived hormonal secretions, small molecule and metabolic cofactor production or through cytokine stress and other inflammatory mechanisms, the permeability of our blood-brain barrier can be significantly impacted. 2. Genetics that may be shared between MS and cardiovascular risks. 3. Smoking increases risk of MS and atherosclerotic disease (ASD). Compared to non-smokers, the risk of MS in smokers is about 50% greater and is correlated with the duration and with the amount of smoking. Smoking also increases the chance of progression of MS. 4. Dyslipidemia Recent observations show that small, dense low-density lipoprotein (LDL) particles (sdLDL) are elevated in autoimmune disease. This lipoprotein subgroup seems to be particularly atherogenic and is a good predictor of significant coronary artery stenosis, further supporting the critical importance of a healthy diet in the treatment plan for MS patients. 5. Hypertension People with multiple sclerosis are 48% more likely to have high blood pressure compared to the general population and to those with other demyelinating diseases. Hypertension can disrupt the BBB and when this barrier is already severely compromised as with MS, the gates are essentially wide open to allow infiltration of autoreactive immune cells into the central nervous system. We generally want our blood pressure to be LESS than 120 systolic and 80 diastolic according to recommendations from the American Heart Association. 6. Exercise What is good for your heart is good for your brain and while MS patients are sometimes less physically active, it’s important to try to incorporate a form of moderate aerobic exercise that can be any activity that increases your heart rate like walking, jogging, or swimming. You just don’t want to overdo it; I tell patients to get to a level where you can talk, but not sing, so you are breathing heavily and may sweat. Ideally aim for at least 150 minutes of total moderate-intensity aerobic activity per week. Stretching is highly recommended for at least 10 minutes per day. It’s the best way to maintain your range of motion and ease symptoms that are related to spasticity (muscle tightness or stiffness or a lack of muscle control). Yoga or tai chi are other great ways to get your stretching in. Strength or resistance training, at least twice a week for 30 minutes each session if possible, helps you maintain and improve your muscle strength while repetitions with lighter weights can aid endurance. You can use machines, dumbbells or resistance bands - just make sure to hit all the muscle groups, but don’t overdo it on your first attempts and risk injury. And of course, always consult your doctor before beginning any new fitness program and make your goals realistic. Just do what you can, when you can! 7. Obesity Some studies in childhood and adolescence showed an association between body mass index (BMI) and the risk of developing multiple sclerosis. Another study found a positive correlation between BMI and Expanded Disability Status Scale (EDSS) score. Patients who were obese had higher EDSS scores at the time of diagnosis, indicating heightened clinical disability and worse disease severity. Investigators also noted that individuals who were obese had higher levels of pro-inflammatory molecules interleukin-6 and leptin as well as lower concentrations of the anti-inflammatory cytokine interleukin-13. They discovered too that triglyceride levels and total to high-density lipoprotein (T/HDL) cholesterol ratios were positively correlated with interleukin-6 concentrations, as obese patients with high triglycerides and elevated T/HDL ratios were also found to have greater levels of interleukin-6. The takeaway? Start now and work with your practitioner to design a health strategy that can effectively and safely address any obesity issues for your own wellbeing and that of your children too.2 For more about childhood obesity see: https://www.cdc.gov/obesity/childhood/defining.html 8. Vitamin D deficiency Numerous studies have linked vitamin D deficiency with both an increased risk and progression of MS and with cardiovascular disease risks as well. Vitamin D is critical for healthy immune cell function and has potent immunomodulatory effects. 9. Obstructive sleep apnea (OSA) increases the risk of stroke and cardiovascular events. MS patients are at a higher risk for OSA and central sleep apnea (CSA ) and both OSA and CSA can contribute to structural neuronal changes leading to central nervous system dysfunction including psychological and physiologic comorbidities. Untreated sleep apnea also is associated with increased risk for dementia, stroke or heart attack. In one study, persons with sleep apnea had a 30% higher risk of heart attack or death than those without apnea. For more about sleep-disordered breathing, see https://www.suzannegazdamd.com/blog/sleep-and-brain-health-can-sleep-disordered-breathing-contribute-to-ad 10. Diabetes Studies have shown higher risk of MS progression in those with diabetes while diabetes also can lead to a five times increased chance of developing Alzheimer’s disease. A growing body of epidemiological and molecular evidence now suggests that a considerable overlap in risk, comorbidity and pathophysiological mechanisms exists across these conditions. Although the brain has long been considered an insulin-independent organ, recent research has shown that insulin actually has significant effects on the brain where it plays a role in maintaining glucose and energy homeostasis. Insulin has two important functions in the brain: controlling food intake and regulating cognitive functions; defects in insulin-signaling in the brain subsequently may contribute to neurodegenerative disorders. We also know that inflammatory processes in the hypothalamus, where insulin receptors are expressed at high density, impaired local signaling systems can result in glucose and energy metabolism disorders due to neural inflammation that affects serum insulin and glucose levels. The hypothalamus is a collection of nuclei with a variety of functions. Many of the important roles of the hypothalamus involve what are known as the two H's: Homeostasis and Hormones. Often this excess neuroinflammation can manifest in corresponding hormone imbalances. And excessive caloric intake and high-fat diets initiate insulin and leptin resistance too by inducing mitochondrial dysfunction and endoplasmic reticulum stress in the hypothalamus. Dr. Dale Bredesen, widely recognized as an expert in the mechanisms of neurodegenerative disease, states "Insulin resistance is the single most important metabolic contributor to Alzheimer’s disease development and progression. It’s vital to ensure your diet does not contribute to insulin resistance." So, if your fasting insulin is: over 4.5 milli-international units per liter (mIU/L); your hemoglobin A1c is over 5.5%; or your fasting glucose over 93 milligrams per deciliter, you are considered to be insulin-resistant. These are critical numbers to know especially if you have diagnosed diabetes or related health conditions including cardiovascular issues. Remember that the human body is not designed to process more than 15 grams of sugar per day, which is far less than you may consume in a 12 ounce soft drink that contains roughly 40 grams. 11. Hyperhomocysteinemia A study published in the Annals of General Psychiatry was conducted to assess the plasma levels of homocysteine in patients with multiple sclerosis (MS) and investigate whether an association with depression exists. Homocysteine is a non-essential sulfur-containing amino acid derived from methionine by demethylation. Vitamins B12 and B6 as well as folate play an important role in the metabolic pathway of homocysteine. A number of recent studies support the notion that homocysteine might cause neuronal damage by triggering oxidative injury and DNA damage. As a result, homocysteine has been implicated in the pathogenesis of numerous neurodegenerative and psychiatric disorders.2 It’s recommended that labs check for this marker as well as MTHFR and B12 levels.4 12. Underlying Infectious triggers The hypothesized mechanism is that an infectious pathogen, possibly with the concurrence of other inducing agents, causes a deregulation of lymphocyte function that leads to an uncontrolled immune reaction in the CNS, resulting in early histological changes with an alteration of the blood-brain barrier that gradually evolve into the more typical CNS lesions. Molecular mimicry between protein sequences from a particular microorganism and structural proteins present in the normal CNS is a possible explanation for autoimmune reactions in MS. Epstein Barr virus (EBV), Human herpesvirus 6 (HH6), chlamydia , fungi, borrelia (associated with Lyme disease as well), and rickettsia all have been implicated. 13. Vascular effects of MS treatment a. Steroids: Systemic glucocorticoids have been associated with an increased risk of cardiovascular diseases including stroke, myocardial infarction, and atrial fibrillation. Nonsteroidal anti-inflammatory drugs (NSAIDs) increase the risk of CVD in the general population. b. Fingolimod, an immunosuppressive drug, has been associated with hypertension, bradycardia and atrioventricular blocks. 14. Stress There is evidence that stress can influence MS onset and its clinical course. Self-reported high stress intensity and weekly/recurring stress were associated with a higher risk of fatal stroke compared with no stress.5 15. EMFs Electromagnetic field (EMF) exposure (manmade sources include cell phone towers, power lines, etc.) can affect structure and function of cardiovascular system and may facilitate myocardial infarction by nuclear changing of cardiomyocytes. Increasing evidence shows that extremely low-frequency electromagnetic fields (ELF-EMFs) stimulation is able to exert a certain action on autoimmunity and immune cells.6 What we must do. Treating any neurological disease including MS requires a personalized protocol to address all layers of underlying factors including: diet; gut-healing; careful laboratory monitoring; identifying any underlying infectious triggers; stress modification; sleep optimization and other efforts designed to promote a healthy lifestyle and strengthen the immune system. Inflammation is at the cornerstone of all disease. Whether you have cancer, heart disease, diabetes, digestive disorders, autoimmune disease, or Alzheimer's, it all starts with inflammation at the cellular level, which leads to either early cell death translating into specific organ or gland disease or into cancer stem cell stimulation, which fuels cancer cell growth and metastasis. We must find a way too for primary care physicians to have the tools and resources to address very early on issues like diabetes and hypertension to potentially improve patient outlooks. Without question, given the adverse impacts of vascular comorbidities on the MS disease trajectory and cardiovascular outcomes in people with MS, vascular risk and vascular comorbidities assessment should become integrated into the care of people with MS. Together, with the medical community working toward a common goal in partnership with our patients, we can achieve better health and futures marked by wellness. In hope and healing, Dr. Suzanne Gazda References: 1 Obrenovich M. (2018). Leaky Gut, Leaky Brain? Microorganisms, 6(4), 107. https://doi.org/10.3390/microorganisms6040107 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6313445/ 2 Munger KL, Bentzen J, Laursen B, Stenager E, Koch-Henriksen N, et al. (2013) Childhood body mass index and multiple sclerosis risk: a long-term cohort study. Mult Scler 19: 1323-1329. 2 Pinhas-Hamiel O, Livne M, Harari G, Achiron A (2015) Prevalence of overweight, obesity and metabolic syndrome components in multiple sclerosis patients with significant disability. Eur J Neurol 22:1275-1279. 2 Stampanoni Bassi M, Iezzi E, Buttari F, et al. Obesity worsens central inflammation and disability in multiple sclerosis [published online ahead of print, 2019 Jun 4]. Multiple Sclerosis Journal. 2019;1352458519853473. doi:10.1177/1352458519853473 3 Herly M, Stengaard-Pedersen K, Horslev-Petersen K, et al. Association between baseline vitamin D metabolite levels and long-term cardiovascular events in patients with rheumatoid arthritis from the CIMESTRA trial: protocol for a cohort study with patient-record evaluated outcomes. BMJ Open 2017; 7:e014816. 3 Sintzel, M. B., Rametta, M., & Reder, A. T. (2018). Vitamin D and Multiple Sclerosis: A Comprehensive Review. Neurology and therapy, 7(1), 59–85. https://doi.org/10.1007/s40120-017-0086-4 4 Triantafyllou, N., Evangelopoulos, M., Kimiskidis, V.K. et al. Increased plasma homocysteine levels in patients with multiple sclerosis and depression. Ann Gen Psychiatry 7, 17 (2008). https://doi.org/10.1186/1744-859X-7-17 5 Thomas Truelsen, Naja Nielsen, Gudrun Boysen, and Morten Grønbæk. Self-Reported Stress and Risk of Stroke, The Copenhagen City Heart Study.13 Mar 2003 https://doi.org/10.1161/01.STR.0000062345.80774.40. Stroke. 2003;34:856–862 Ret. 2020 from https://www.ahajournals.org/doi/full/10.1161/01.str.0000062345.80774.40 5 Artemiadis A, K, Anagnostouli M, C, Alexopoulos E, C: Stress as a Risk Factor for Multiple Sclerosis Onset or Relapse: A Systematic Review. Neuroepidemiology 2011;36:109-120. doi: 10.1159/000323953 https://www.karger.com/Article/PDF/323953 6 Guerriero, F., & Ricevuti, G. (2016). Extremely low frequency electromagnetic fields stimulation modulates autoimmunity and immune responses: a possible immuno-modulatory therapeutic effect in neurodegenerative diseases. Neural regeneration research, 11(12), 1888–1895. https://doi.org/10.4103/1673-5374.195277 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5270416/ Additional resources: Scalfari, A., Knappertz, V., Cutter, G., Goodin, D. S., Ashton, R., & Ebers, G. C. (2013). Mortality in patients with multiple sclerosis. Neurology, 81(2), 184–192. https://doi.org/10.1212/WNL.0b013e31829a3388 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3770174/ Marrie RA, Cutter G, Tyry T. Substantial adverse association of visual and vascular comorbidities on visual disability in multiple sclerosis. Multiple Sclerosis Journal. 2011;17(12):1464-1471. doi:10.1177/1352458511414041
0 Comments
Your comment will be posted after it is approved.
Leave a Reply. |
AuthorDr. Suzanne Gazda, Integrative Neurology Archives
February 2024
Categories |
 RSS Feed
RSS Feed