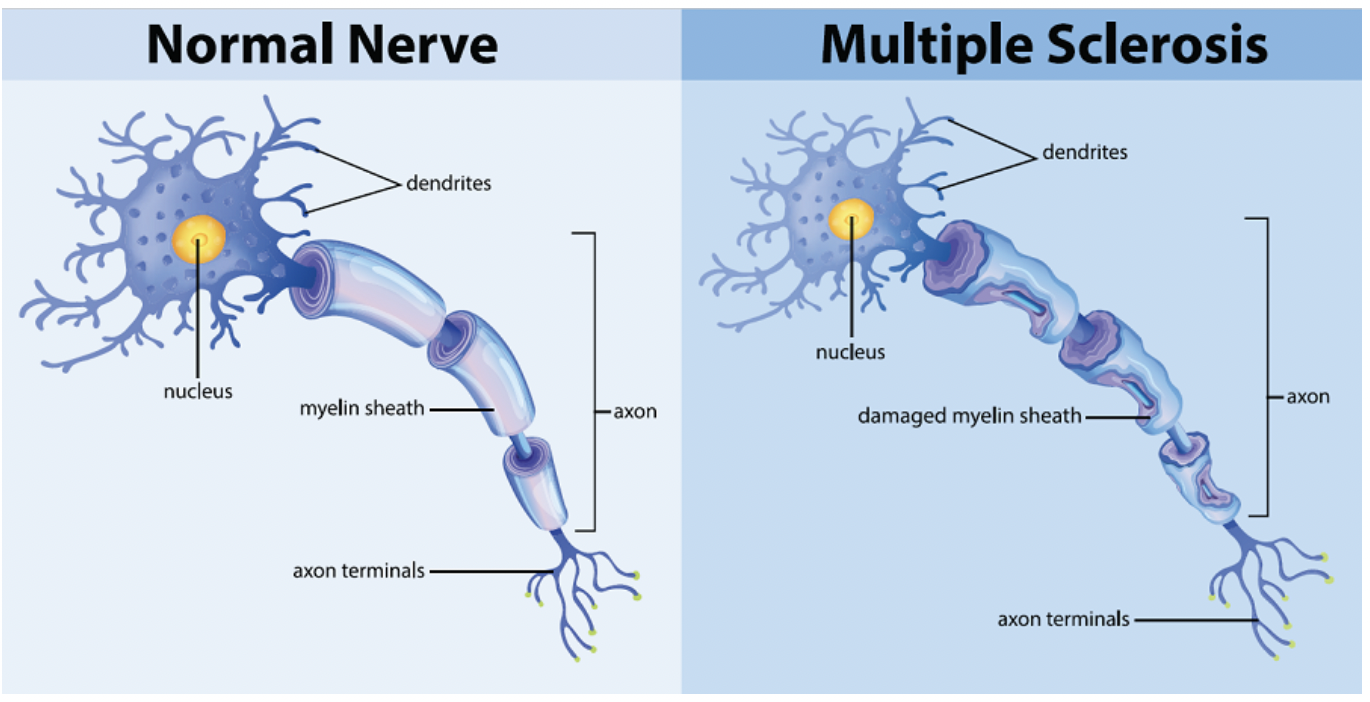
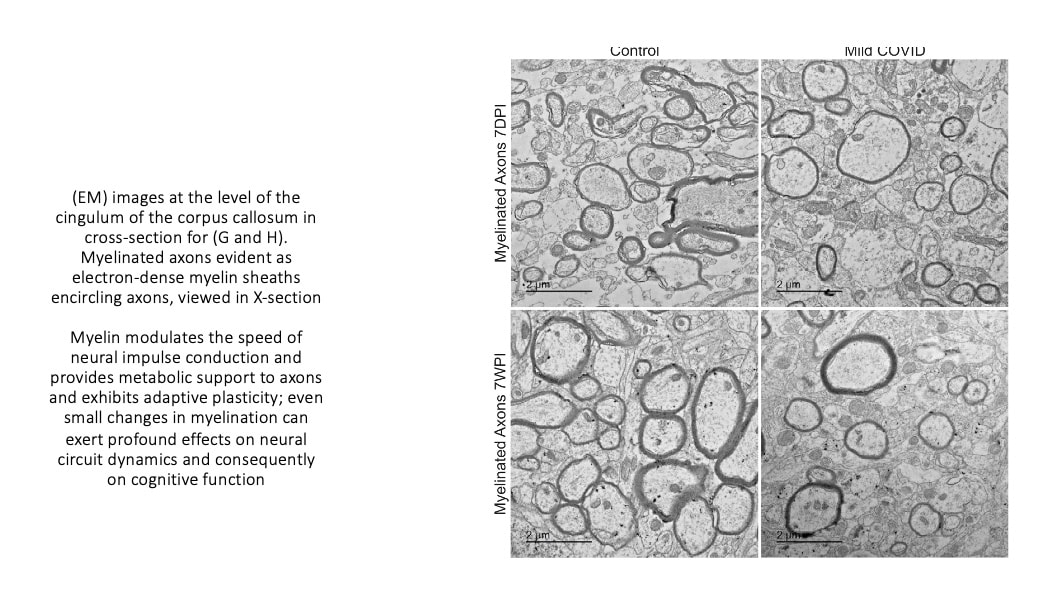
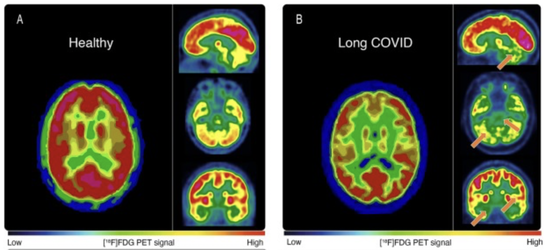
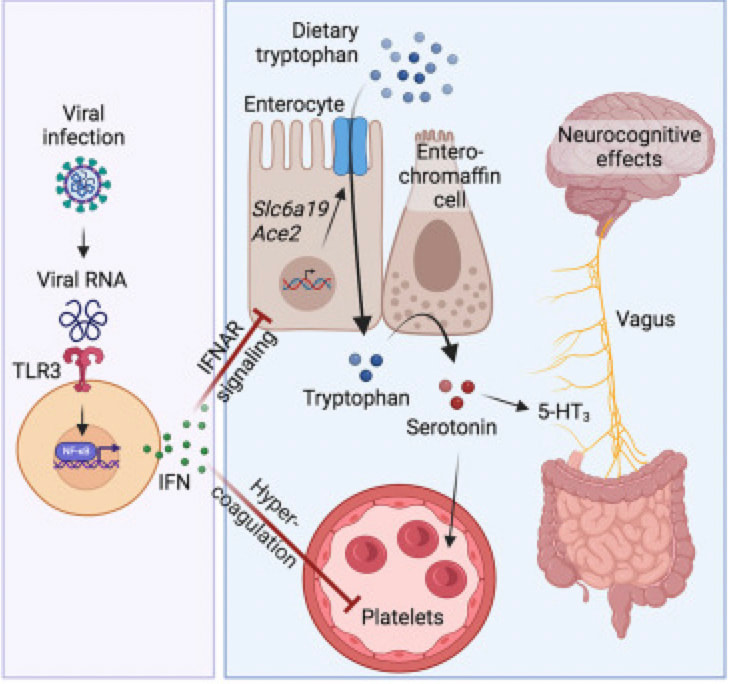
COVID 19 and increased physical disability progression in MS: A Report from the Front Lines.10/25/2023 Multiple sclerosis (MS) is an autoimmune inflammatory illness that affects the central nervous system (CNS) when the body's immune system attacks and destroys myelin, the fatty substance that coats and protects nerve fibers in the brain and spinal cord. It is characterized by demyelination and varying degrees of axonal loss, which is an important component of the pathology of MS. Our practice has since 2020 and especially since 2021 seen a distinct rise in both MS progression and what I can only call a tsunami of various new demyelinating diagnoses. But what exactly is fueling these events? Numerous case reports (l have listed a few in references below) connect post-COVID infection or post vaccine with new onset or worsening demyelinating disease. A recent presentation at The European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS) confirms what we have observed in our own MS patients. The presentation looked closely at a 2022 study that included 231 participants with MS. Results showed that of the 88 subjects with COVID-19 and 143 without those post infection experienced an increased risk of higher expanded disability status scale (EDSS) score, and subsequent increased risk of disability progression. Researchers compared EDSS changes in participants from January 2020 to October 2021. By now, most of us have had COVID one or more times, and most adults in the US received the COVID vaccine/vaccines. Both have made our medical practices more challenging than ever before. I have written about how lingering viral fragments and in particular spike protein and mRNA, originating either post infection or post vaccine, are strong drivers of molecular mimicry and autoimmune disease. In a 2023 study published in Nature, the research detailed how molecular mimicry of the SARS-CoV-2 viral fragments and MS proteins such as myelin share an eerily similar homology. And study authors noted that molecular mimicry is " likely unique to SARS-CoV-2 among the major human coronaviruses”. Several investigations, including one by Monje et al and a study by Fernanez-Castaneda, showed damage to oligodendrocytes, which are the cells that make myelin as well as a dramatic reduction of myelinated axons. In addition, a study by Huang in 2022 showed persistent white matter changes in recovered COVID-19 patients at the one-year follow up. In this diagram presented in a study by Michelle Monje and team, we can see the drop out of myelinated axons which are the lines of communication in the brain. These myelinated axons are in the brain’s white matter and act like a superhighway system to relay information quickly. Here you can see this “highway” has a few less roads to travel down. Besides molecular mimicry, other factors driving this dramatic shift could include: 1. Alteration of glutamate pathways In MS and EAE or experimental autoimmune encephalomyelitis, which is an animal model of brain inflammation, there are excess glutamate levels and multiple abnormalities in glutamate- degrading enzymes, glutamate transporters, glutamate receptors, and glutamate signaling. Glutamate is an excitatory neurotransmitter in the brain that fuels neuroinflammation. While glutamate is the main excitatory neurotransmitter, GABA is the main inhibitory neurotransmitter. Inhibitory neurotransmitters decrease the likelihood that a nerve impulse will fire; GABA normally inhibits brain activity, enabling you to relax. When your GABA levels are low, your brain essentially gets stuck in the “on” position and you can find yourself in a continuous state of being anxious, overstimulated, and overwhelmed. Much as the accelerator and brakes in your car work together to control speed, GABA puts the brakes on brain activity to counter glutamate’s accelerative effects. A recent study looked at how astrocytes, which are key responders to neuroinflammation, can have reduced capacity to take up glutamate. The hypometabolism pattern observed in long COVID patients with brain fog using fluorodeoxyglucose (FDG)-positron emission tomography (PET) or FDG-PET scans might be a signature of astrocyte-related glutamatergic dysregulation. 2. Alteration of the microbiome post infection and post vaccine The brain and gut are inextricably linked in a critical gut-brain connection. Research has shown that gut dysbiosis or the imbalance of the gut microbiome in MS could certainly affect disease progression. Infections with the COVID-19 virus have been shown to damage the gut microbiome and are associated with compromised integrity of the gut’s mucous layer, causing gut dysbiosis which is a microbiome imbalance. The COVID-19 mRNA vaccine is linked to reduced biodiversity in the microbiome. Dr. Sabine Hazan, a gastroenterologist and founder and CEO of a research genetic sequencing laboratory, ProgenaBiome, has also observed drastic losses of Bifidobacteria, which are a group of bacteria called probiotics that normally live in your intestines and stomach. They help your body perform essential functions such as digestion and warding off harmful bacteria. 3. Low serotonin MS has been shown to be associated with low serotonin levels. Serotonin is a chemical that carries messages between nerve cells in the brain and throughout your body, playing a key role in such body functions as mood, sleep, and digestion. Low serotonin levels can have a negative effect on immunomodulation. Researchers at the University of Pennsylvania suggest that serotonin reduction is triggered by remnants of viral RNA lingering in the gut which increased an immune response with high amounts of interferon which limits the absorption of tryptophan, the precursor to serotonin. Many microbes in the gut use dietary nutrients to make neurotransmitters. It is estimated that respectively, 95 percent of serotonin and 50 percent of dopamine originate in the gut. 4. Vascular inflammation MS and vascular disease go hand in hand, which is why we always strive to reduce the relevant risk factors. There is now little controversy that COVID can lead to vascular disease. In this well designed study, researchers showed strong correlations between neuroinflammation and vascular dysfunction. The results were not surprising given that the brain, which uses 15-20 percent of the body's total circulating blood supply, is uniquely vulnerable to disruptions to vascular health. 5. Alteration of the Glymphatic system The glymphatic system is relatively newly discovered macroscopic waste clearance system that utilizes a unique system of perivascular channels, formed by astroglial cells, to promote efficient elimination of soluble proteins and metabolites from the central nervous system. If your glymphatic system is altered it can adversely impact the waste removal processes; as noted in one study, this imbalance between the production and clearance of proteins within the brain, one of which being Amyloid Beta (AB) may contribute to the development of Alzheimer’s disease. In multiple sclerosis, an impairment of the glymphatic system may contribute to pathology accrual in the central nervous system and disability progression. Recent evidence has shown the glymphatic abnormalities have been described in several neurodegenerative conditions, including Alzheimer's as well as Parkinson's disease. Additional research has revealed as well that long COVID and concurrent brain fog could be a result of damage or disruption of glymphatics and failure in cleansing system of wastes and proinflammatory cytokines. 6. Alteration of the BBB and high levels of neuroinflammation.
I have previously explained how spike protein has been shown to linger post COVID and after a COVID vaccine. Direct exposure of the brain's primary immune cells or microglia to spike protein causes high levels of inflammatory cytokines and microglial cell activation. Spike protein independently acts like a pathogen to induce neuroinflammatory processes via pattern recognition receptor engagement. And as we have discussed many, many times in the past, inflammation lies at the core of every neuroimmune and autoimmune disease. For additional insights, see: The dramatic rise in autoimmune disease post COVID and post vaccine. https://www.suzannegazdamd.com/blog---long-covid/the-dramatic-rise-in-autoimmune-disease-post-covid-and-post-vaccine New onset autoimmune issues post COVID and post vaccine. https://www.suzannegazdamd.com/blog---long-covid/new-onset-autoimmune-issues-post-covid-and-post-vaccine Spike protein on its own will disrupt the blood-brain barrier (BBB) – and what drives neuroinflammation and harms the BBB will increase the risk of neurodegeneration and potential progression of related conditions including multiple sclerosis. As all clinicians have experienced, there is no question that the events of 2020 and ensuing years have brought enormous changes to medicine and our perspective on disease origins. Perhaps most important, this situation has emphasized further the need to continue pursuing all lines of inquiry to identify the most appropriate treatments for all patients. In hope and healing, Dr. Suzanne Gazda References: Hillert J, et al. The effect of COVID-19 infection on physical disability progression in MS patients at Karolinska University Hospital. Presented at: ECTRIMS 2023; Oct. 11-13, 2023. https://www.healio.com/news/neurology/20231019/covid19-linked-to-increased-risk-of-physical-disability-progression-in-ms Lake CM, Breen JJ. Sequence similarity between SARS-CoV-2 nucleocapsid and multiple sclerosis-associated proteins provides insight into viral neuropathogenesis following infection. Sci Rep. 2023;13(1):389. Published 2023 Jan 8. doi:10.1038/s41598-022-27348-8 https://pubmed.ncbi.nlm.nih.gov/36617594/ Horowitz, T. et al. Brain fog in long COVID: A glutamatergic hypothesis with astrocyte dysfunction accounting for brain PET glucose hypometabolism. Medical Hypotheses. Nov 2023. https://www.sciencedirect.com/science/article/pii/S0306987723001822 Cornell University Newsroom. Study Finds Bacterial Toxin May Trigger Multiple Sclerosis Onset and Relapse. Weill Cornell Medicine, Office of Public Affairs. March 2023. https://news.weill.cornell.edu/news/2023/03/study-finds-bacterial-toxin-may-trigger-multiple-sclerosis-onset-and-relapse#:~:text=A%20specific%20toxin%2Dproducing%20gut,Cornell%20Medicine%20and%20NewYork%2DPresbyterian. Leung JSM. Interaction between gut microbiota and COVID-19 and its vaccines. World J Gastroenterol. 2022;28(40):5801-5806. doi:10.3748/wjg.v28.i40.5801. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9639653/ Monje, M. et al. The neurobiology of long COVID. Cell. 2022. https://www.cell.com/neuron/pdf/S0896-6273(22)00910-2.pdf Fernández-Castañeda A, Lu P, Geraghty AC, et al. Mild respiratory SARS-CoV-2 infection can cause multi-lineage cellular dysregulation and myelin loss in the brain. Preprint. bioRxiv. 2022;2022.01.07.475453. Published 2022 Jan 10. doi:10.1101/2022.01.07.475453 Huang S, Zhou Z, Yang D, et al. Persistent white matter changes in recovered COVID-19 patients at the 1-year follow-up. Brain. 2022;145(5):1830-1838. doi:10.1093/brain/awab435 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8754808/ Levite, M. et al. Glutamate, T cells and multiple sclerosis. J Neural Transm (Vienna). 2017;124(7):775-798. doi:10.1007/s00702-016-1661-z Altieri C, Speranza B, Corbo MR, Sinigaglia M, Bevilacqua A. Gut-Microbiota, and Multiple Sclerosis: Background, Evidence, and Perspectives. Nutrients. 2023;15(4):942. Published 2023 Feb 14. doi:10.3390/nu15040942 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9965298/ Leung JSM. Interaction between gut microbiota and COVID-19 and its vaccines. World J Gastroenterol. 2022;28(40):5801-5806. doi:10.3748/wjg.v28.i40.5801. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9639653/ Hazan S, Stollman N, Bozkurt HS, et al Lost microbes of COVID-19: Bifidobacterium, Faecalibacterium depletion and decreased microbiome diversity associated with SARS-CoV-2 infection severity. BMJ Open Gastroenterology 2022;9:e000871. doi: 10.1136/bmjgast-2022-000871 San Hernandez AM, Singh C, Valero DJ, et al. Multiple Sclerosis and Serotonin: Potential Therapeutic Applications. Cureus. 2020;12(11):e11293. Published 2020 Nov 2. doi:10.7759/cureus.11293 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7707915/ Wong AC, Devason AS, Umana IC, et al. Serotonin reduction in post-acute sequelae of viral infection [published online ahead of print, 2023 Oct 9]. Cell. 2023;S0092-8674(23)01034-6. doi:10.1016/j.cell.2023.09.013. https://pubmed.ncbi.nlm.nih.gov/37848036/ Minagar A, Jy W, Jimenez JJ, Alexander JS. Multiple sclerosis as a vascular disease. Neurol Res. 2006;28(3):230-235. doi:10.1179/016164106X98080 Feng, J. How Are Multiple Sclerosis and Hardening of the Arteries (Atherosclerosis) Linked? Newswise Articles, Oshner Health. 2023. https://www.newswise.com/articles/how-are-multiple-sclerosis-and-hardening-of-the-arteries-atherosclerosis-linked Zanini G, Selleri V, Roncati L, et al. Vascular "Long COVID": A New Vessel Disease? [published online ahead of print, 2023 Jan 18]. Angiology. 2023;33197231153204. doi:10.1177/00033197231153204 Jessen NA, Munk AS, Lundgaard I, Nedergaard M. The Glymphatic System: A Beginner's Guide. Neurochem Res. 2015;40(12):2583-2599. doi:10.1007/s11064-015-1581-6 Eyre, B. Guest Blog – How does the brain clean itself? And is this linked to Alzheimer’s? Dementia Researcher. 2021. https://www.dementiaresearcher.nihr.ac.uk/guest-blog-how-does-the-brain-clean-itself-and-is-this-linked-to-alzheimers/ Rocca, M. Glymphatic system impairment may contribute to multiple sclerosis pathology. European Academy of Neurology. 2022. https://www.ean.org/research/resources/neurology-updates/detail/glymphatic-system-impairment-may-contribute-to-multiple-sclerosis-pathology#:~:text=In%20multiple%20sclerosis%2C%20an%20impairment,nervous%20system%20and%20disability%20progression. Bukhari, M. et al. Glymphatics and brain fog – the post-COVID-19 phenomenon. Biomedica. 2022. https://biomedicapk.com/10.51441/BioMedica/5-648 Olajide OA, Iwuanyanwu VU, Adegbola OD, Al-Hindawi AA. SARS-CoV-2 Spike Glycoprotein S1 Induces Neuroinflammation in BV-2 Microglia. Mol Neurobiol. 2022;59(1):445-458. doi:10.1007/s12035-021-02593-6.https://pubmed.ncbi.nlm.nih.gov/34709564/ Gazda, S. Small fiber neuropathy after COVID infection and vaccination. suzannegazdamd.com. 2023. https://www.suzannegazdamd.com/blog---long-covid/small-fiber-neuropathy-after-covid-infection-and-vaccination DeOre BJ, Tran KA, Andrews AM, Ramirez SH, Galie PA. SARS-CoV-2 Spike Protein Disrupts Blood-Brain Barrier Integrity via RhoA Activation. J Neuroimmune Pharmacol. 2021;16(4):722-728. doi:10.1007/s11481-021-10029-0. https://pubmed.ncbi.nlm.nih.gov/34687399/ Additional references: Alluqmani, M. et al. New Onset Multiple Sclerosis Post-COVID-19 Vaccination and Correlation with Possible Predictors in a Case-Control Study. Cureus. May 2023 Shabani, Z. Demyelination as a result of an immune response in patients with COVID-19. Acta Neurol Belg. 2021. Batruch, O. et al. Covid-19 vaccination can induce multiple sclerosis via cross-reactive CD4+ T cells recognizing SARS-CoV-2 spike protein and myelin peptides. Multiple Sclerosis Journal ; 28(3 Supplement):776, 2022. Janarious, A. et al. Severe Presentation of Neuromyelitis Optica with Positive Antibody Following COVID-19 Vaccination. Neurology. 2022. Fujikawa, P. et al. Neuromyelitis Optica in a Healthy Female After Severe Acute Respiratory Syndrome Coronavirus 2 mRNA-1273 Vaccine (Moderna). Cureus. 2021. Caliskan, I. et al. A Case With New-Onset Neuromyelitis Optica Spectrum Disorder Following COVID-19 mRNA BNT162b2 Vaccination. Neurologist (Turkey). 2022. Lohman, L. et al. Severe disease exacerbation after mRNA COVID-19 vaccination unmasks suspected multiple sclerosis as neuromyelitis optica spectrum disorder: a case report. BMC Neurology. 2022 Anamnart, C. et al. Newly diagnosed neuromyelitis optica spectrum disorders following vaccination: Case report and systematic review. MS and Related Disorders. Feb 2022 Fragoso, Y. New relapse of multiple sclerosis and neuromyelitis optica as a potential adverse event of AstraZeneca AZD1222 vaccination for COVID-19. Mult Scler Relat Disord. 2022. Paybast, S. et al. Watch out for neuromyelitis optica spectrum disorder onset or clinical relapse after COVID-19 vaccination: What neurologists need to know? Mult Scler Relat Disord. 2022. Harel, T. et al. New onset or relapsing neuromyelitis optica temporally associated with SARS-CoV-2 infection and COVID-19 vaccination: a systematic review. Frontiers Neurology. 2023. Toljan K, Amin M, Kunchok A, Ontaneda D. New diagnosis of multiple sclerosis in the setting of mRNA COVID-19 vaccine exposure. J Neuroimmunol. 2022;362:577785. doi:10.1016/j.jneuroim.2021.577785 Harahsheh, E. et al. Aquaporin-4 IgG neuromyelitis optica spectrum disorder onset after Covid-19 vaccination: Systematic review. J of Neuroimmunology. Dec 2022 Francis, A. et al. Acute Inflammatory Diseases of the Central Nervous System After SARS-CoV-2 Vaccination. Neuroimmunology & Neuroinflammation. Jan 2023. Schultz, H. et al. First manifestation of multiple sclerosis after immunization with the Pfizer-BioNTech COVID-19 vaccine. J Neurol [Epub ahead of print] 2021. Etemadifar M, Sigari AA, Sedaghat N, Salari M, Nouri H. Acute relapse and poor immunization following COVID-19 vaccination in a rituximab-treated multiple sclerosis patient. Hum Vaccin Immunother. 2021;17(10):3481-3483. doi:10.1080/21645515.2021.1928463 Maniscalco GT, Manzo V, Di Battista ME, et al. Severe Multiple Sclerosis Relapse After COVID-19 Vaccination: A Case Report. Front Neurol. 2021;12:721502. Published 2021 Aug 10. doi:10.3389/fneur.2021.721502 Apostolidis, S. et al. Altered cellular and humoral immune responses following SARS-CoV-2 mRNA vaccination in patients with multiple sclerosis on anti-CD20 therapy. MedRxiv. 2021. Youn T, Yang H. Cytotoxic Lesion of the Corpus Callosum (CLOCCs) after SARS-CoV-2 mRNA Vaccination. J Korean Med Sci. 2021;36(31):e228. Published 2021 Aug 9. doi:10.3346/jkms.2021.36.e228 Seyed Ahadi M, Ghadiri F, Ahraian MA, Naser Moghadasi A. Acute attack in a patient with multiple sclerosis 2 days after COVID vaccination: a case report. Acta Neurol Belg. 2023;123(1):253-254. doi:10.1007/s13760-021-01775-2 Khayat-Khoei M, Bhattacharyya S, Katz J, et al. COVID-19 mRNA vaccination leading to CNS inflammation: a case series. J Neurol. 2022;269(3):1093-1106. doi:10.1007/s00415-021-10780-7 Maramattom B.V. et al. Myelin Oligodendrocyte Glycoprotein-Associated Disorders Post-ChAdOx1 Vaccination. Cureus. 2022;14(3):e23197. Published 2022 Mar 15. doi:10.7759/cureus.23197 Rinaldi, V. et al. CNS inflammatory demyelinating events after COVID-19 vaccines: A case series and systematic review. Front Neurology. Dec 2022. Nabizadeh, F. et al. Multiple sclerosis relapse after COVID-19 vaccination: A case report-based systematic review. J Clin Neurosci. 2022.
0 Comments
Your comment will be posted after it is approved.
Leave a Reply. |
AuthorSuzanne Gazda M.D. Neurologist Archives
January 2024
Categories |


 RSS Feed
RSS Feed