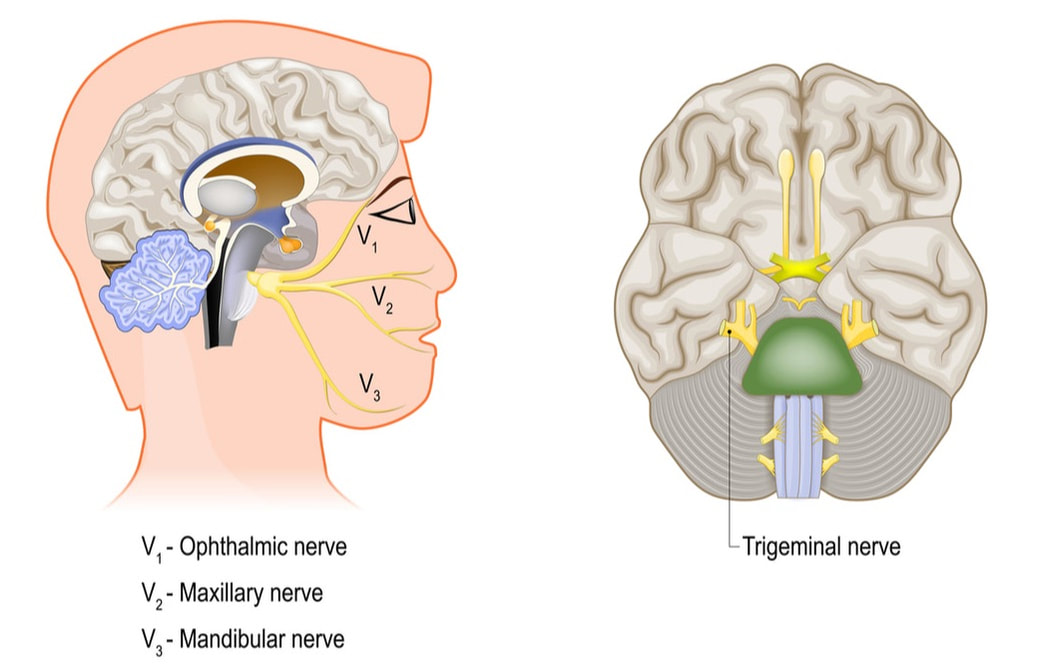
Neuroplasticity, the brain’s ability to change itself in response to the environment, has shown us that that the brain isn’t as rigidly hardwired or entirely immutable. Through novel modalities using light, sound, electricity and motion, neuroplasticity can help our brains adapt to either circumstances of injury or disease and truly heal. And now, we also have advancements in technology that offer hope for patients suffering from traumatic brain injuries (TBI) and certain symptoms in multiple sclerosis (MS). PoNS®, the portable neuromodulation stimulator device and TBI. A non-invasive system designed to encourage new neural connections, the PoNS device sits on the surface of a patient’s tongue and delivers mild, gentle electrical impulses to the surface of the tongue. In patients experiencing mild to moderate mobility issues post-TBI, studies have evidenced structural changes leading to some functional recovery in patients experiencing mild to moderate mobility issues post-TBI.1 The impulses excite the neural network flowing to the brain. This neural activity, combined with therapeutic activities, is believed to enable “neuroplasticity” which may restore lost function. Note that while PoNS is available for TBI patients in Canada, the U.S. is still reviewing its application for use in this particular diagnosis. PoNS is now approved for MS protocols. For MS patients ages 22 and older, the device recently was authorized by the Food and Drug Administration (FDA) to address gait-related deficits, one of the most common symptoms of this neurodegenerative disorder. The device is intended to be used by prescription only as a complementary adjunct to a supervised therapeutic exercise program. In a randomized, controlled, double-blind trial involving 20 patients with MS-related gait deficits, ten patients used the PoNS device and ten used a “sham” control device that did not deliver any stimulation. Compared with the control group, the PoNS group achieved "statistically significant and clinically significant" improvement in their Dynamic Gait Index (DGI) score at the end of the study (14 weeks), the FDA said in its recent news release. How exactly does the device work? As a portable and non-implanted device, PoNS delivers mild neuromuscular electrical stimulation to the dorsal surface or top of the patient's tongue. It consists of a controller and a mouthpiece that are connected to each other by a cord. During a visit with a physical therapist or relevant clinician, patients retain a mouthpiece held lightly in place by the lips and teeth and wear the control unit around the neck. The controller sends signals to the mouthpiece while receptors on the tongue transmit millions of neural impulses to the brain through natural pathways. Additionally, the therapist can connect the control unit to a computer and view usage data via the proprietary developed software. This data provides important information on how to improve each patient's complementary physical therapy program by identifying potentially problematic areas of missed or shortened sessions. The stimuli delivered under the tongue trigger the trigeminal and facial nerves to excite a natural flow of neural impulses to the brainstem or cerebellum that subsequently promotes changes in targeted brain structures. PoNS vs. the pons area of the brain. Latin for “bridge,” the pons region is the largest part of the brainstem, located above the medulla and below the midbrain. It is a group of nerves that function as a connection between the cerebrum and cerebellum. The earliest work with translingual neurostimulation (TLNS) has been in patients with TBIs. Results from pilot studies of TLNS treatment of patients after mild to moderate TBI suggest that, in the absence of identifiable tissue damage, a combination of neurostimulation and rehabilitation that is both targeted and challenging will induce neuroplastic changes (e.g. brain regions of pons, brainstem, and cerebellum), reduce symptoms, and begin normalising function. One study showed there are significant changes in gray matter volume (GMV). The gray matter contains most of the brain's neuronal cell bodies and includes regions of the brain involved in muscle control as well as sensory perception such as seeing and hearing, memory, emotions, speech, decision making, and impulse control. Could there be other positive outcomes? For example, if this device is paired with cognitive therapy, is there the potential for benefits in cognitive function? At present there are additional trials underway to research these considerations. What, if any, are the risks?
Some participants had reported a mild tingling sensation that lasted from several minutes up to an hour after the stimulus was turned off and some noticed excess salivation as a result of the presence of the PoNS in mouth. These side effects can be attenuated by adjusting the intensity of the stimulus. Makers of the PoNS device note it should not be used by patients with penetrating brain injuries, neurodegenerative diseases, oral health problems, chronic infectious diseases, unmanaged hypertension or diabetes, pacemakers and/or a history of seizures. And as always, patients or their families are encouraged to discuss any questions or concerns with their physician or prescribing clinician. The big question will be…for how long do the improvements last? With neuroplasticity training, we do have to consider that when we stop training the brain, the beneficial effects may not endure. But just as with any protocol or therapeutic approach, both time and patient follow up are needed to determine the duration and levels of efficacy. Certainly, this is an exciting innovation that could help improve symptoms and quality of life for many neurology patients and we will continue to watch with great interest future studies and real-time applications! With hope and healing, Dr. Suzanne Gazda 1 The European Medical Journal, EMJ Reviews https://www.emjreviews.com/radiology/article/translingual-neural-stimulation-with-the-portable-neuromodulation-stimulator-pons-induces-structural-changes-leading-to-functional-recovery-in-patients-with-mild-to-moderate-traumatic-brain-in/
4 Comments
Joanna Hokanson D.D.S.
5/7/2021 10:27:18 am
Hi Dr. G-
Reply
Jacqueline Haller
6/27/2021 06:40:09 am
After 20 years of MS treated from 2000 to 2006 with Rebif(efficient to that year) and then Avonex(not efficient to calm down my lesions' number increase) i found this website ww w. kykuyuhealthclinic. com just 3 months ago, and their current natural health tech to help curb/manage it. It has made a tremendous difference for me I had improved walking balance, muscle strength and improved vision, always thankful for nature that helps in managing these terrible diseases.
Reply
wa nye steve
9/19/2022 11:51:31 pm
I never thought i could smile and be in a happy marriage again if not for the help of DR.Moses Buba. I got the doctors Emails:([email protected] ) on the internet and i emailed him, and he got back to me with some encouraging words, he got me some herbs cream which i use for just 8 days and i began to feel the enlargement of my penis, and without surgery. This went on for a little period of about 10 days and to my surprise my wife keeps screaming that she love my big dick now. Now my wife no longer cheat on me, and my penis is now about 10.5 inches long on erection and off course very large round. And now my wife uses breasts, hips and bums enlargement. I and my wife are very happy for the help rendered to me by DR.Moses Buba, and i want to say a big thanks to Doctor for the help. You can contact the Doctor now on his Email:( [email protected] ) Am thankful to the doctor for helping me.
Reply
Mavis Wanczyk
7/20/2023 07:26:41 pm
Being the winner of a multi-million dollar lottery certainly will be a life-changing event for almost every single lottery winner. My name is Mavis Wanczyk from Chicopee, Massachusetts, the famous PowerBall lottery winner of $758 million (£591m). I know many people would wonder how I had won the lottery. Would you believe me if I told you that I did it with spell casting? I met this famous spell caster known as Doctor Odunga and he was the one who did it for me. As shocking as it was to me, my famous comment to the press was “ I’m going to go and hide in my bed.” Never did I believe that Doctor Odunga made me wealthy overnight. If you want to have your chance of winning and becoming very wealthy just like me, contact Doctor Odunga at [email protected] OR WHATS-APP HIM at +2348167159012 and you will be lucky. Thanks for reading and hope to see you at the top
Reply
Your comment will be posted after it is approved.
Leave a Reply. |
AuthorSuzanne Gazda M.D. Archives
July 2024
Categories |

 RSS Feed
RSS Feed