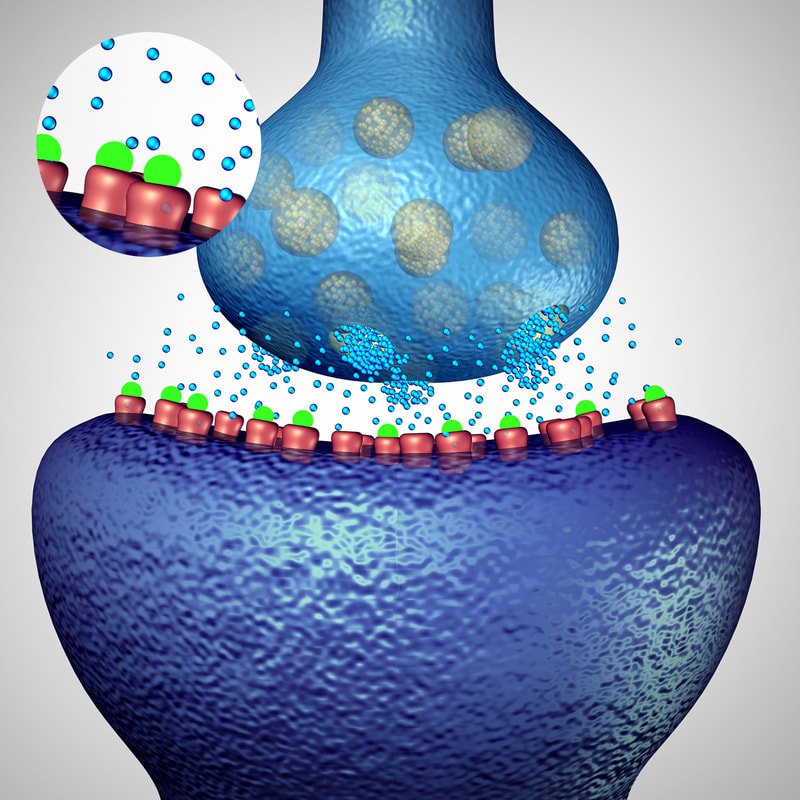
Typically it’s confined to our brain cells and is released only when it needs to send a “message” and then is immediately retracted back into the cell. But in the presence of trauma or neurodegenerative disease, vast levels of glutamate may be expelled and subsequently cause a variety of symptoms and disease activity.
Memantine has been employed in treatment for Alzheimer’s disease and seeks to suppress the excess release of glutamate with the intent to improve outcomes in patients with Alzheimer’s. While it has been shown to prevent functional disability and cognitive decline, its long-term benefit in treating this disease appears to be minimal – and by “long term” that could mean just one year’s time. The implications for use in reducing some MS symptoms. But the question remained as to whether this glutamate suppressor could be of use in another neurodegenerative disease such as MS. Specifically, several clinical trials sought to determine if memantne could reduce spasticity, fatigue and disability, as well as the related cognitive impairment suffered by patients. The latter is especially problematic as it affects approximately 45-65% of MS patients and causes considerable disability and lifestyle burdens. Damage to the descending motor tracts brought about by autoimmune processes and subsequent neuroplasticity seen in MS is thought to cause disinhibition of motor reflexes leading to spasticity. Early studies involving rodent models of MS also suggest the role of glutamate toxicity in spasticity. MS-related fatigue is understood to be related to axonal injury secondary to glutamate toxicity as manifested by the reduction in N-acetylaspartate levels seen in multivoxel spectroscopic studies.1 Glutamate has been implicated in the autoimmune demyelination that we see in our MS patients as increased levels of the neurotransmitter glutamate have been identified in their brains, which lowers the levels of N-acetylaspartate (NAA) — a process that likely leads to the loss of brain volume. These findings indicate that glutamate might be a driver of neuronal cell death and disease progression in MS, and a potential target of new therapies.2 However, this trial and several others, which have been carried out to look at cognitive function, also showed no significant benefit. We do know that cognitive function is very difficult to objectively measure, particularly in a short-term study. So while more studies are warranted, the opportunity to reduce excessive glutamate in active neurological disease is one that we should continue to consider for those patients with associated symptoms. As part of a treatment protocol… I don’t think at this time that we should be prepared to eliminate memantine from our list of therapeutic options, especially given what we now know about the over-activation of the glutamatergic system and glutamate toxicity in neurodegeneration. Remembering that whichever form is used it must be slowly titrated to assess patient tolerance, we generally follow these dosing practices:
For ways to naturally reduce or control glutamate levels, see: https://www.suzannegazdamd.com/blog/the-relationship-between-glutamate-and-gaba Be sure to talk to your physician or neurology specialist if you have questions or concerns about your treatment regimen and, of course, please feel free to reach out to our office if you would like to schedule a visit. In hope and healing, Dr. Suzanne Gazda References: 1 Mehta, L. R., McDermott, M. P., Goodman, A. D., & Schwid, S. R. (2010). A randomized trial of memantine as treatment for spasticity in multiple sclerosis. Multiple sclerosis (Houndmills, Basingstoke, England), 16(2), 248–251. https://doi.org/10.1177/1352458509355462 2 Azevedo, C. J., Kornak, J., Chu, P., Sampat, M., Okuda, D. T., Cree, B. A., Nelson, S. J., Hauser, S. L., & Pelletier, D. (2014). In vivo evidence of glutamate toxicity in multiple sclerosis. Annals of neurology, 76(2), 269–278. https://doi.org/10.1002/ana.24202
2 Comments
wanye steve
9/19/2022 11:56:56 pm
I never thought i could smile and be in a happy marriage again if not for the help of DR.Moses Buba. I got the doctors Emails:([email protected] ) on the internet and i emailed him, and he got back to me with some encouraging words, he got me some herbs cream which i use for just 8 days and i began to feel the enlargement of my penis, and without surgery. This went on for a little period of about 10 days and to my surprise my wife keeps screaming that she love my big dick now. Now my wife no longer cheat on me, and my penis is now about 10.5 inches long on erection and off course very large round. And now my wife uses breasts, hips and bums enlargement. I and my wife are very happy for the help rendered to me by DR.Moses Buba, and i want to say a big thanks to Doctor for the help. You can contact the Doctor now on his Email:( [email protected] ) Am thankful to the doctor for helping me.
Reply
Mavis Wanczyk
7/20/2023 07:24:04 pm
Being the winner of a multi-million dollar lottery certainly will be a life-changing event for almost every single lottery winner. My name is Mavis Wanczyk from Chicopee, Massachusetts, the famous PowerBall lottery winner of $758 million (£591m). I know many people would wonder how I had won the lottery. Would you believe me if I told you that I did it with spell casting? I met this famous spell caster known as Doctor Odunga and he was the one who did it for me. As shocking as it was to me, my famous comment to the press was “ I’m going to go and hide in my bed.” Never did I believe that Doctor Odunga made me wealthy overnight. If you want to have your chance of winning and becoming very wealthy just like me, contact Doctor Odunga at [email protected] OR WHATS-APP HIM at +2348167159012 and you will be lucky. Thanks for reading and hope to see you at the top
Reply
Your comment will be posted after it is approved.
Leave a Reply. |
AuthorSuzanne Gazda M.D. Archives
July 2024
Categories |
 RSS Feed
RSS Feed